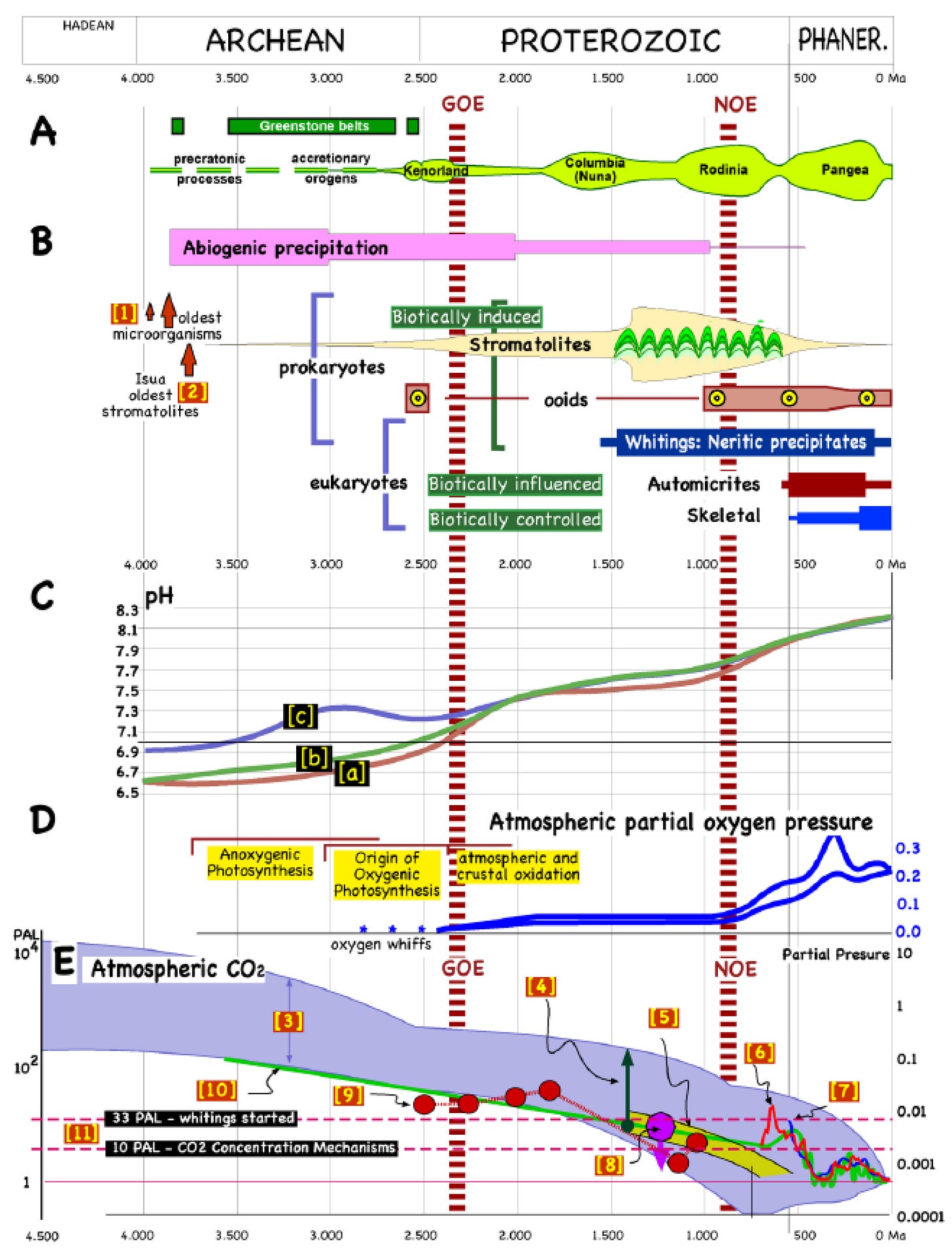
Image by Pomar et al., 2022 — see at the end of the post for the caption. This image generated a post that you may find a little hard to digest. But the nutrient cycles of the ecosphere are such a fascinating subject that I couldn’t avoid writing about them, and I hope I succeeded in conveying at least some of this fascination to you.
When I saw the image above, it reminded me of the periodic table of the elements, a piece of knowledge that we take for granted today, but that was a major innovation when it appeared for the first time in the 1860s.
It was the result of several contributions, but the most remarkable one was provided by Dmitri Mendeleyev, who was bold enough to propose that the regularities in the table indicated that there had to be elements that hadn’t been discovered yet. From then on, we speak of “Mendeleev’s table of the elements,” the cornerstone of all we know in chemistry.
The impressive thing about the table of elements is that there is a certain order; a certain logic in the universe. Our first attempts to systematize knowledge in new fields often lead to mistakes and haphazard ideas. But, eventually, the logic comes out. There are reasons for what we see.
It is happening right now for our knowledge of the history of Earth; Gaia’s life story. After at least a couple of centuries of work, a logic is appearing. We are seeing Gaia as a living being that was born, grew, changed, experienced catastrophes, recovered, grew again, and now is perhaps at her climax, already seeing the start of its terminal decline that will last for at least a few hundred million years.
So, we have the impressive image above, provided by Luis Pomar in a recent paper. If you are not familiar with these things, you may get lost in the arcane terminology used. For instance, “NOE” stands for “Neoproterozoic oxygenation event”, while “GOE” stands for “Great Oxygenation Event” — but the former was greater than the latter! And I bet you have never heard of “ooids,” (“egg stones”), spherical aggregations of sedimented calcium carbonate. But now you know, and there they are, appearing perhaps 2.5 billion years ago.
It is the way things are: Gaia’s life history is much more complicated than the nicely ordered rows and columns of the periodic table of the elements. I suggest that you peruse this graph for a while to get a sense of how these enormous changes unfolded over this enormous span of time. I was impressed by the sequence of the continental plates: Kenorland, Columbia, Rodinia, and Pangea (and there were small continents even before Kenorland). I didn’t know that the Ocean pH (“C” box in the image) increased gradually over billions of years, but it is the logical consequence of the gradual decline of CO2 concentrations in the atmosphere (this is the core subject of the paper). Note, anyway, that some elements of the graph are highly uncertain, and, for instance, the oxygen concentration curve is probably much flatter than the one reported in section D. Also, the data on the concentration of atmospheric CO2 (sec. E) may change as new studies appear.
It is easy to get lost in this table; it is so incredibly packed with data and events. In any case, if you have the time to go through the whole paper, you’ll learn many things that go well beyond the simplified view of the homeostasis of Earth’s climate we often have. An element that’s often not taken into consideration is that of “biogenic carbonates,” a fundamental part of the planetary carbon cycle. The gradual increase in solar irradiation has led to lower CO2 concentration in the atmosphere in order to maintain the temperature homeostasis of the system. That, in turn, led to the idea that modern plants are CO2-starved — which is probably wrong (but it will take another long post to explain why).
What you may not have known (I didn’t before reading this paper) is that the availability of phosphate as a nutrient has also been going down with the Eons. Yes, because phosphorous doesn’t have a stable gaseous molecule, and it is available only as phosphate ions in water (PO43−). But this availability depends on the pH, and for high pHs, (more basic), phosphates tend to precipitate as solid calcium compounds. It is a good thing that phosphates are not easily soluble in water, otherwise things would be bad for your teeth and your bones. In any case, CO2 tends to make the water acidic, and the gradual decline of CO2 concentrations in the air made waters more basic. Hence, phosphate ions became less available to living creatures: modern life on Earth is became PO43−— starved. Still today, phosphates are a major component of the fertilizers used in agriculture.Not so complicated once you get the knack of it, but it is a series of concatenations that need some time to be understood.
The idea proposed in the paper I am discussing here is that life coped with these unfavorable conditions by developing what they call the “PEM,” the Phosphate Extraction Mechanism. It is a cycle generated by the diurnal shift of photosynthesis and respiration that acts as a pump to precipitate and dissolve carbonates, making phosphates available for living creatures during the acidic phase. Here is how the authors describe the mechanism:
Photosynthesis typically involves daytime CO2 uptake and O2 release predominating over respiration; at night, respiration consumes O2 and releases CO2. The result is strong daily variation in both pH and dissolved O2 concentrations in aquatic environments. In warm-water environments in which alkalinity is relatively high and PO43− concentrations are minimal, these variations have the potential to promote precipitation of CaCO3 and associated adsorption of PO43− in daylight when photosynthesis is active. At night, lower pH and oxygen availability can promote some CaCO3 dissolution and PO43− desorption. The combination of calcification and PO43− extraction occurs in photosynthesizing cyanobacteria, as well as in a diverse array of calcifying nanophytoplankton and calcareous macroalgae, indicating that this process allows both prokaryotic and eukaryotic photosynthetic organisms to thrive in warm, alkaline, oligotrophic waters. Concentrations of both atmospheric CO2 and surface-ocean PO43−, which are essential for biological productivity, have declined by at least 1–2 orders of magnitude over Earth history. The Phosphate Extraction Mechanism, associated with photosynthetically induced calcification, has played a major role in the production and accumulation of carbonates throughout much of the Proterozoic and the Phanerozoic.
Complicated story, right? Indeed. Gaia is a female goddess, and, as a lady of great experience, she is sometimes difficult to understand for us, mere humans. But surely she is fascinating in all her manifestations. (image generated by Dezgo.com)
_________________________________________________________________
Caption of Figure 2 (from Pomar et al.). Carbonate precipitation nodes, genetic macroevolution rates, and estimates of oxygen and CO2 in the atmosphere. (A): Supercontinents and crustal growth; precratonic, accretionary orogens and supercontinents assembly (redrawn from multiple sources). (B): Carbonate precipitation modes through Earth’s history (multiple sources). [1]: Oldest fossils [18]. [2]: Isua oldest stromatolites [19]. (C): pH evolution from [20]; [a]: nominal model, in which the median Archean surface temperature is slightly higher than modern surface temperatures. Archean land fraction was anywhere between 10% and 75% of modern land fraction; [b]: no Archean land endmember scenario; [c]: model with assumed 100 ppm Proterozoic methane and 1% Archean methane levels. (D): Maximum and minimum estimates of atmospheric partial oxygen pressure (after [21]). (E): Estimates of atmospheric CO2 concentration; [3] from [22], upper and lower boundaries reflect average surface temperature for an ice-free (20 °C) and ice covered (5°) Earth; [4] Acritarch isotopic composition [23]; [5] C-isotope reservoir modeling [24]; [6] Phanerozoic GEOCARB III models [25]; [7] Royer compilation [26]; [8] from [27]; [9] paleosoil mass balances [28]; [10] from [29]; [11] Picoplanktonic whiting and partial sheath calcification commenced 1400–1300 Ma ago (33 CO2 PAL), and cyanobacteria CCMs were induced when pCO2 = 10 PAL [30]. PAL: present atmospheric level; GOE: the Great Oxygenation Event; NOE: Neoproterozoic oxygenation event. (This figure is an original compilation and interpretation of data by L. Pomar, based upon numerous sources).





Yeah no thrusting of lands happening anywhere. The oxygenation period!? There is something amiss. Like when they dig these organic material out of mud and it biolumineses a myriad of colours until the O2 ? Or C gets into it. Where does this come from?
Thank you, Ugo. I look at that wonderful graphic and try to insert a "You are Here" arrow, but it is very small and points to the little point where Atmospheric CO2 touches the X-axis "today".
Another interesting phenomenon has been how the Himalayas got thrust up by India hitting Eurasia, and how acid-rain on the calcium of the Himalayas ends up as calcium carbonate at the bottom of the Indian Ocean. "Bug" or "feature"?
"Mother Nature's (bastard) Son"